STIP1 (1-543, His-tag) Human Protein
Other products for "STIP1"
Specifications
| Product Data | |
| Species | Human |
| Expression Host | E. coli |
| Expression cDNA Clone or AA Sequence |
MGSSHHHHHH SSGLVPRGSH MEQVNELKEK GNKALSVGNI DDALQCYSEA IKLDPHNHVL YSNRSAAYAK KGDYQKAYED GCKTVDLKPD WGKGYSRKAA ALEFLNRFEE AKRTYEEGLK HEANNPQLKE GLQNMEARLA ERKFMNPFNM PNLYQKLESD PRTRTLLSDP TYRELIEQLR NKPSDLGTKL QDPRIMTTLS VLLGVDLGSM DEEEEIATPP PPPPPKKETK PEPMEEDLPE NKKQALKEKE LGNDAYKKKD FDTALKHYDK AKELDPTNMT YITNQAAVYF EKGDYNKCRE LCEKAIEVGR ENREDYRQIA KAYARIGNSY FKEEKYKDAI HFYNKSLAEH RTPDVLKKCQ QAEKILKEQE RLAYINPDLA LEEKNKGNEC FQKGDYPQAM KHYTEAIKRN PKDAKLYSNR AACYTKLLEF QLALKDCEEC IQLEPTFIKG YTRKAAALEA MKDYTKAMDV YQKALDLDSS CKEAADGYQR CMMAQYNRHD SPEDVKRRAM ADPEVQQIMS DPAMRLILEQ MQKDPQALSE HLKNPVIAQK IQKLMDVGLI AIR
|
| Tag | His-tag |
| Predicted MW | 64.8 kDa |
| Concentration | lot specific |
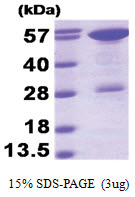
| Purity | >90% by SDS - PAGE |
| Buffer | Presentation State: Purified State: Liquid purified protein Buffer System: 20 mM Tris-HCl buffer (pH 8.0) containing 1 mM DTT, 20% glycerol, 1 mM EDTA, 0.2 mM PMSF |
| Preparation | Liquid purified protein |
| Protein Description | Recombinant Human STI1, fused to His-tag at N-terminus, was expressed in E.coli and purified by using conventional chromatography techniques. |
| Storage | Store undiluted at 2-8°C for up to two weeks or (in aliquots) at -20°C or -70°C for longer. Avoid repeated freezing and thawing. |
| Stability | Shelf life: one year from despatch. |
| Reference Data | |
| RefSeq | NP_001269581 |
| Locus ID | 10963 |
| UniProt ID | P31948 |
| Cytogenetics | 11q13.1 |
| Synonyms | HEL-S-94n; HOP; IEF-SSP-3521; P60; STI1; STI1L |
| Summary | STIP1 is an adaptor protein that coordinates the functions of HSP70 (see HSPA1A; MIM 140550) and HSP90 (see HSP90AA1; MIM 140571) in protein folding. It is thought to assist in the transfer of proteins from HSP70 to HSP90 by binding both HSP90 and substrate-bound HSP70. STIP1 also stimulates the ATPase activity of HSP70 and inhibits the ATPase activity of HSP90, suggesting that it regulates both the conformations and ATPase cycles of these chaperones (Song and Masison, 2005 [PubMed 16100115]). [supplied by OMIM, Jul 2009] |
| Protein Families | Stem cell - Pluripotency |
| Protein Pathways | Prion diseases |
Documents
| FAQs |
Resources
Recombinant Protein Resources |
{0} Product Review(s)
0 Product Review(s)
Submit review
Be the first one to submit a review
Product Citations
*Delivery time may vary from web posted schedule. Occasional delays may occur due to unforeseen
complexities in the preparation of your product. International customers may expect an additional 1-2 weeks
in shipping.






























































































































































































































































 Germany
Germany
 Japan
Japan
 United Kingdom
United Kingdom
 China
China