Tissue FFPE Sections, Breast
CAT#: CS804569
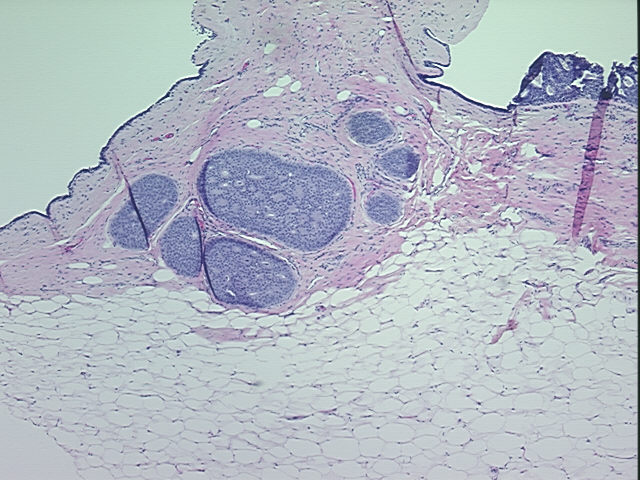
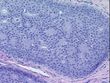
Tissue Sections (5x5um), FFPE; Breast; Carcinoma in situ of breast, ductal
USD 248.00
2 Weeks*
USD 331.00
USD 179.00
Specifications
| Product Data | |
| Disease State | Cancer |
| Diagnosis Category | Breast Cancer |
| Tissue | Breast |
| Frozen Sample ID | PA00003A9C |
| Case ID | CU0000005213 |
| Tissue of Origin | Breast |
| Site of Finding | Breast |
| Appearance | T |
| Sample Diagnosis (from Pathology Verification) | Carcinoma in situ of breast, ductal |
| Normal | 80% |
| Lesional | 0% |
| Tumor | 15% |
| Tumor Hypo/Acellular Stroma | 0% |
| Tumor Hypercellular Stroma | 5% |
| Necrosis | 0% |
| Pathology Verification notes from H&E review | no invasive carcinoma present, only ductal carcinoma in-situ, cribriform pattern; 80% normal component comprising 5% glandular epithelium, 5% stroma, 70% fat |
| CASE Diagnosis (from medical center path report) | Adenocarcinoma of breast, ductal |
| Age | 67 |
| Gender | Female |
| Tumor Grade | Nottingham G2: 6-7 points Intermediate combined grade (moderately favorable) |
| Minimum Stage Grouping | IIA |
| T (Extent of Primary Tumor) | T1a |
| N (Lymph node metastasis) | N1a |
| M (Distant metastasis) | MX |
| Diagnostic Test Results | Progesterone receptor ~ PR ! by IHC,Positive; Estrogen receptor ~ ER ! by IHC,Positive; HER2 ~ ERBB2 ~ Neu ! by IHC,Positive, weak |
Documents
| SDS |
{0} Product Review(s)
Be the first one to submit a review






























































































































































































































































 Germany
Germany
 Japan
Japan
 United Kingdom
United Kingdom
 China
China